As siRNA programs advance toward larger-scale manufacture, the effect of backbone modifications on product consistency is receiving greater attention. In Codexis’ presentation at TIDES USA 2025, the team explored how thiophosphate stereochemistry contributes to these challenges and how improved control may support more reliable siRNA production.
Why is stereochemistry important in RNA therapeutics?
In siRNA therapeutics, certain backbone modifications introduce structural features that can influence a molecule’s performance. One key modification is the introduction of thiophosphate linkages, created when a non-bridging oxygen atom in the phosphate backbone is replaced with sulfur. This substitution introduces chirality at the phosphorus, resulting in the creation of diastereomers in Rp and Sp configurations (Figure 1).
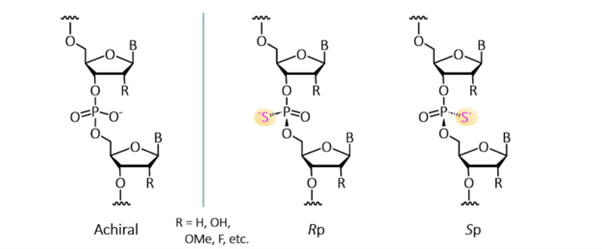
Figure 1. Substitution of a non-bridging oxygen with sulfur in the phosphate linkage converts an achiral phosphate into a chiral thiophosphate, creating two possible configurations, Rp and Sp.
Each diastereomer has distinct properties. Evidence shows that when stereochemistry is not controlled during synthesis, the resulting mixtures of Rp and Sp products impact the properties of the siRNA therapeutic, potentially affecting its stability, activity and toxicity [1,2,3].
The stereochemical and operational limits of SPOS
Solid phase oligonucleotide synthesis (SPOS) is the primary method used to produce therapeutic siRNAs. In this process, each nucleotide is added through a controlled series of reactions on a solid support, and any thiophosphate linkage formed during these steps is created through standard chemical coupling. As these reactions do not distinguish between the two possible configurations at the chiral phosphorus center, SPOS produces a mixture of Rp and Sp diastereomers. This mixture becomes more complex as additional thiophosphate linkages are introduced, making it difficult to predict or influence the stereochemical profile of the final molecule.
As programs advance toward larger-scale manufacture, the operational constraints of SPOS also become more visible. The method depends on high volumes of solvents and reagents, generates substantial chemical waste that requires careful handling and disposal, and relies on energy and equipment-intensive operations that add complexity as batch sizes grow. These factors make it challenging to manage stereochemical outcomes while meeting the performance and sustainability expectations of modern RNA manufacturing.
Scalable RNA manufacturing with ECO Synthesis
The ECO Synthesis® Manufacturing Platform offers an alternative approach to siRNA manufacturing that addresses scalability and sustainability constraints.
Instead of solid support chemistry, ECO Synthesis uses an enzyme-enabled cycle in which an engineered polymerase carries out the extension step and a phosphatase prepares the molecule for the next addition. The process takes place in an aqueous environment, and the overall workflow reduces the need for organic solvents while offering a more adaptable path for scale-up as programs advance (Figure 2).
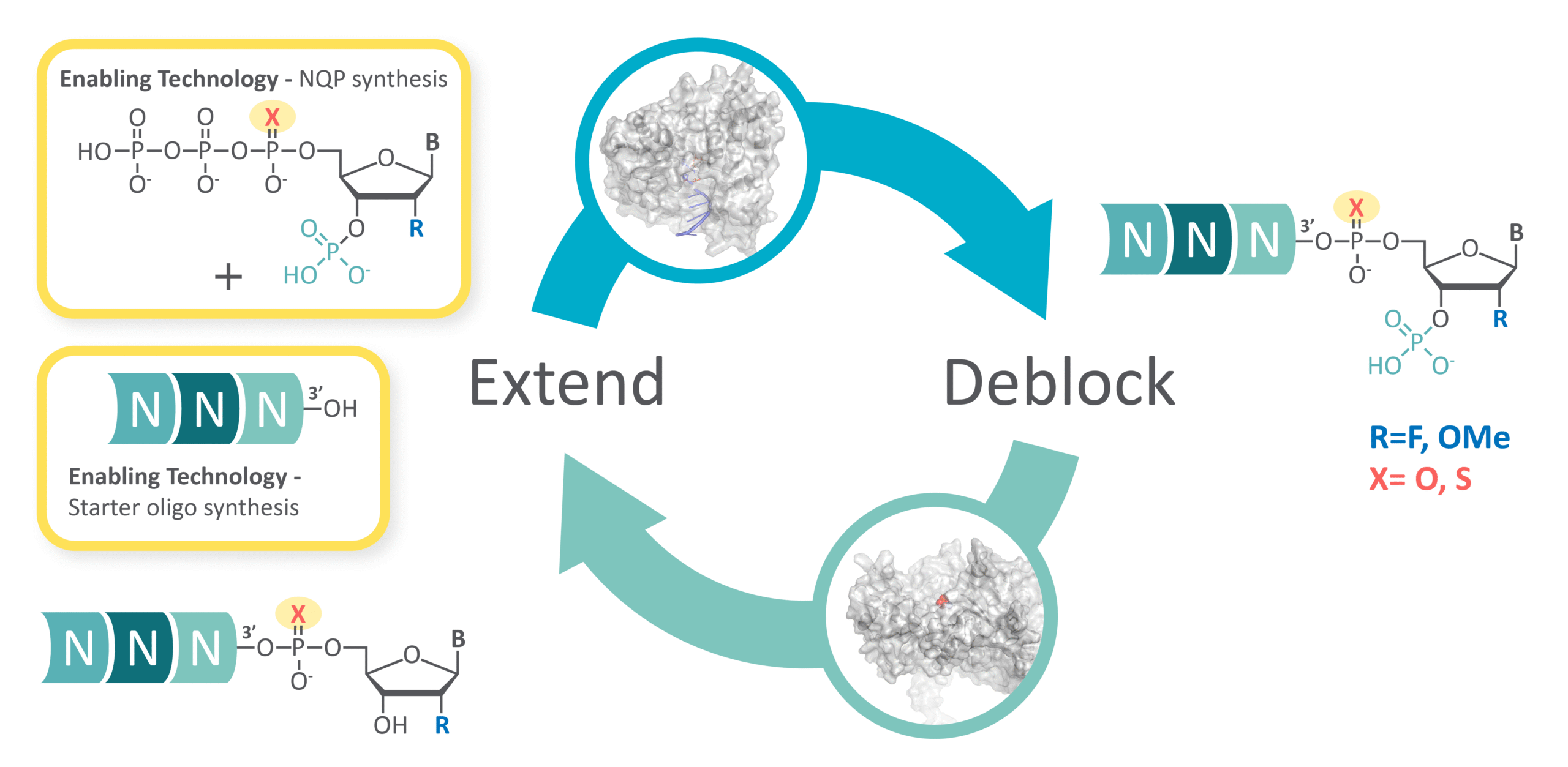
Figure 2. Overview of the ECO Synthesis enzymatic cycle. An engineered polymerase incorporates the next nucleotide, extending the siRNA by one position, and a phosphatase removes the blocking group to prepare the molecule for the following addition.
How does ECO Synthesis control stereochemistry?
In addition to offering a more sustainable and scalable option for RNA manufacturing, ECO Synthesis allows greater influence over the stereochemistry introduced at thiophosphate linkages. This control comes from the engineered polymerases developed specifically to guide how each linkage is formed. During nucleotide addition, the polymerase sets the three-dimensional arrangement around the phosphorus center, and with the appropriate substrate, it can introduce a defined configuration at that position.
This means ECO Synthesis can steer whether an Rp or Sp configuration is incorporated at a given linkage, allowing development teams to work toward a more consistent stereochemical profile across the siRNA. As programs advance toward later stages, this capability supports more confident evaluation of stability, activity and toxicity across key performance attributes. It also provides a level of precision that is difficult to achieve through SPOS, where both configurations form without distinction.
Exploring stereochemical control at TIDES USA
In Codexis’ presentation at TIDES USA 2025, the team demonstrated how ECO Synthesis uses engineered polymerases to direct the introduction of Rp or Sp configurations during thiophosphate bond formation. Through controlled experiments, the session showed how different polymerase and substrate combinations influence stereochemistry at defined linkage positions within an siRNA.
The presentation also offered a side-by-side comparison of stereochemical profiles generated through SPOS and ECO Synthesis, illustrating how enzyme-enabled steps can support more consistent outcomes across modified linkages.
Watch the full presentation to learn more about how Codexis can help you evolve beyond traditional RNA chemistry with ECO Synthesis.
References
- Sakamuri et al. ChemBioChem 2020. 21, 1304-1308. https://doi.org/10.1002/cbic.201900630
- Jahns et al. Nuc. Acids Res. 2022. 50,3 1221-1240. https://doi.org/10.1093/nar/gkab544
- Liu et al. Nuc. Acids Res. 2023. 51, 9, 4126-4147. https://doi.org/10.1093/nar/gkad268
Date Published:
17 February 2026
Discover the Expertise Powering Possibility
Speak to our experts to learn how our platforms deliver smarter science, faster timelines, and greater confidence. Let’s talk.

Related Content
4 February 2026
5 mins read time
Accelerating Scalable and Sustainable siRNA Manufacturing with Ligation-based Synthesis
Want to see more blogs?






We're Paving Your Path to the Future
You own the product and we own the path. Together, we’ll take it to market clearly, cleanly and without surprises.